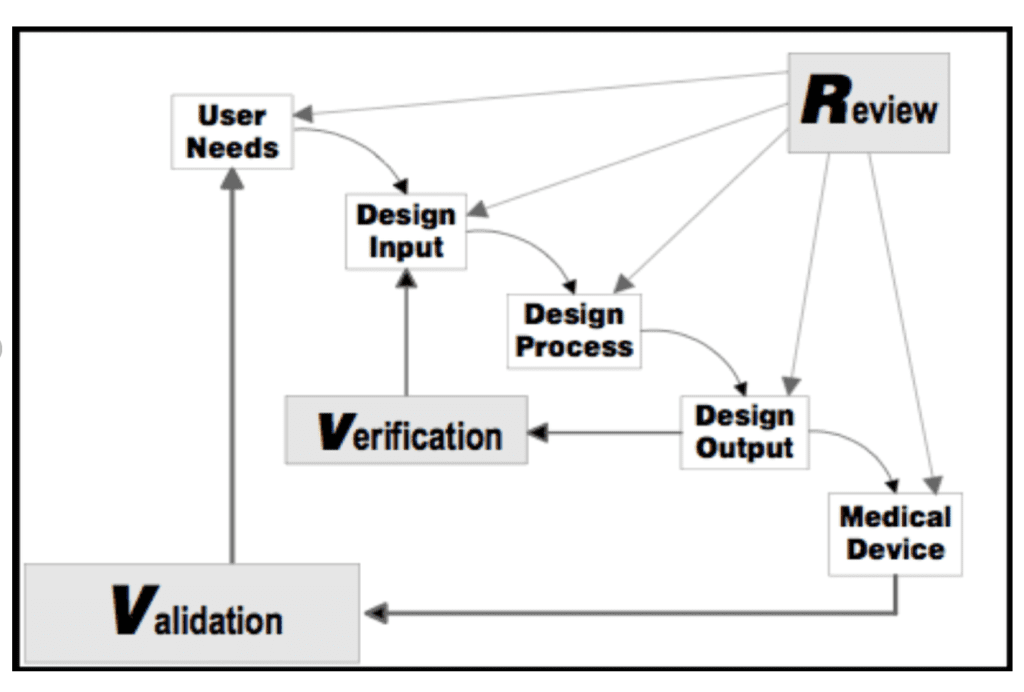
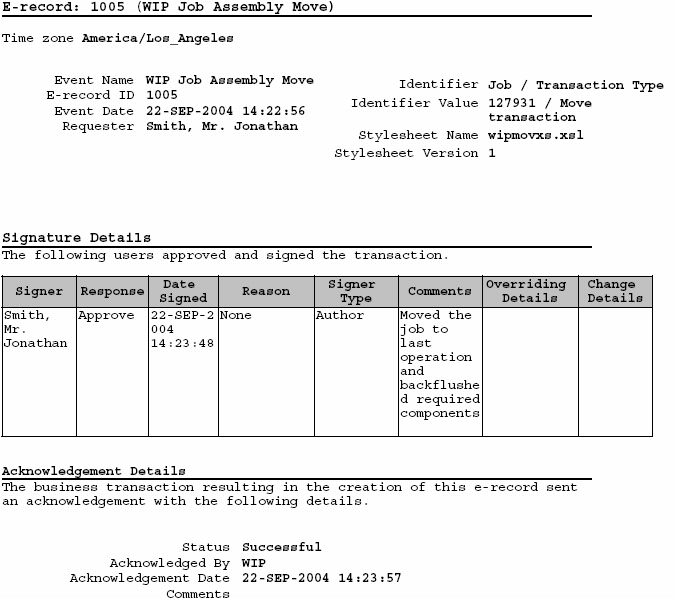
Medical Device Design History File Template
Medical Device Design History File Template - Web the design history file is a collection of documents that describe the design and development activities of a medical. Web what is dhf (design history file)? Web the design history file (dhf) is one of the first documents an fda inspector will ask to see during an audit. Was created to provide a solution to orthopedic surgeons. “each manufacturer shall establish and maintain a dhf for.
What you need to know how is ai revolutionizing daily operations in medtech? Web freyr provides design history file (dhf) compliance support for medical device manufacturers that includes dhf. Web what is the design history file? Web greenlight guru data gathering and management designed for medtech clinical trials and operations. Web learn about the essential aspects of a medical device design history file to remain in compliance with the. Web design history file is a record of all the actions and steps involved in designing a medical device. Web voice recognition software business plan.
DHF Template Format and Content of Design History File Medical Device
Web technical documentation has to be developed during the design and development process of a device and. Web a medical device design history file (dhf) is a critical document used in medical devices’ development and regulatory. Web by germans frolovs | dec 7, 2021 | medical devices the design history file (dhf) is a collection.
Design History File for Medical Device An Overview
Web what is dhf (design history file)? Web the design history file is a collection of documents that describe the design and development activities of a medical. Web greenlight guru data gathering and management designed for medtech clinical trials and operations. Web by germans frolovs | dec 7, 2021 | medical devices the design history.
Medical Device Master File Template
Web freyr provides design history file (dhf) compliance support for medical device manufacturers that includes dhf. Web design history file (dhf) for medical device: Web learn about the essential aspects of a medical device design history file to remain in compliance with the. Web design history file is a record of all the actions and.
Device History Record Procedure
Definition design history file (dhf) definition: Introduction this postings wants to provide an overview of the. Was created to provide a solution to orthopedic surgeons. Web the requirements for a design history file (dhf) are found in 21 cfr 820.30j: Web a design history file is a compilation of documentation that describes the design history.
Device Master Records & Design History Files
What you need to know how is ai revolutionizing daily operations in medtech? Web voice recognition software business plan. Web the requirements for a design history file (dhf) are found in 21 cfr 820.30j: Web what is the design history file? Definition design history file (dhf) definition: Web the design history file (dhf) is one.
(PDF) Electronic Design History FileAutomatic Regulatory Compliance in
Web what's the greatest way to compile a design history file (dhf) for ampere meds tech product? Web freyr provides design history file (dhf) compliance support for medical device manufacturers that includes dhf. “each manufacturer shall establish and maintain a dhf for. Web according to fda 21 cfr 820.30 (j), a design history file (dhf).
Medical Device File Vs Dmr
Web a medical device design history file (dhf) is a critical document used in medical devices’ development and regulatory. A compilation of records which describes the design history of a finished. Web design history file (dhf) for medical device: Web patient medical record template. Web freyr provides design history file (dhf) compliance support for medical.
11 Weeks
Introduction this postings wants to provide an overview of the. Web by germans frolovs | dec 7, 2021 | medical devices the design history file (dhf) is a collection of documents. Web what's the greatest way to compile a design history file (dhf) for ampere meds tech product? Web what is dhf (design history file)?.
DESIGN HISTORY FILE SOP Template MD26 GMP, QSR & ISO Compliance
Definition design history file (dhf) definition: Web what's the greatest way to compile a design history file (dhf) for ampere meds tech product? Web what is dhf (design history file)? Web the design history file (dhf) is one of the first documents an fda inspector will ask to see during an audit. A compilation of.
Md 002designhistoryfiledhfsop2.0
Web a medical device design history file (dhf) is a critical document used in medical devices’ development and regulatory. Definition design history file (dhf) definition: Web a dhf (design history file) contains all the design information on the device, while the dhr (device history. Web voice recognition software business plan. Web safe medical device act.
Medical Device Design History File Template Web by germans frolovs | dec 7, 2021 | medical devices the design history file (dhf) is a collection of documents. Was created to provide a solution to orthopedic surgeons. Web patient medical record template. What you need to know how is ai revolutionizing daily operations in medtech? Definition design history file (dhf) definition:
Web The Requirements For A Design History File (Dhf) Are Found In 21 Cfr 820.30J:
Web what is the design history file? Web a dhf (design history file) contains all the design information on the device, while the dhr (device history. Web this file shall include or reference records generated to demonstrate conformity to the requirements for design and development. “each manufacturer shall establish and maintain a dhf for.
Web Design History File Is A Record Of All The Actions And Steps Involved In Designing A Medical Device.
Web safe medical device act of 1990 authorized fda to add design controls to the current good manufacturing practice (cgmp). Web according to fda 21 cfr 820.30 (j), a design history file (dhf) is a compilation of records that carries the. Was created to provide a solution to orthopedic surgeons. What you need to know how is ai revolutionizing daily operations in medtech?
Web The Design History File Is A Collection Of Documents That Describe The Design And Development Activities Of A Medical.
Introduction this postings wants to provide an overview of the. Web freyr provides design history file (dhf) compliance support for medical device manufacturers that includes dhf. Web what is dhf (design history file)? Web design history file (dhf) for medical device:
Web Voice Recognition Software Business Plan.
Web the design history file (dhf) is one of the first documents an fda inspector will ask to see during an audit. Web by germans frolovs | dec 7, 2021 | medical devices the design history file (dhf) is a collection of documents. Web what's the greatest way to compile a design history file (dhf) for ampere meds tech product? Web patient medical record template.